/lithiumatom-56a12c335f9b58b7d0bcc103.jpg)
In 1855, Robert Bunsen and Augustus Matthiessen produced large quantities of the metal by electrolysis of lithium chloride. The element was not isolated until 1821, when William Thomas Brand performed Brande also described pure salts of lithium, such as the chloride, and performed an estimate of its atomic weight. Nevertheless, the neurological effect of the lithium ion Li + makes some lithium salts useful as a class of mood stabilizing drugs. Trace amounts of lithium are present in the oceans and in some organisms, though the element serves no apparent biological function in humans. Lithium occurs in a number of pegmatitic minerals, but is also commonly obtained from brines and clays on a commercial scale, lithium metal is isolated electrolytically from a mixture of lithium chloride and potassium chloride. Lithium is the 33rd most abundant element on Earth, but due to its high reactivity only appears naturally in the form of compounds. The reasons for its disappearance and the processes by which new lithium is created continue to be important matters of study in astronomy. For this reason, lithium metal is typically stored under the cover of oil.Īccording to theory, Lithium (mostly 7Li) was one of the few elements synthesized in the Big Bang, although its quantity has vastly decreased.

Like all alkali metals, lithium is highly reactive, corroding quickly in moist air to form a black tarnish. Under standard conditions, it is the lightest metal and the least dense solid element. It is a soft alkali metal with a silver-white color. This fact has key implications for the building up of the periodic table of elements.Template:Infobox lithium Lithium ( pronounced /ˈlɪθiəm/) is a chemical element with the symbol Li and atomic number 3. The ordering of the electrons in the ground state of multielectron atoms, starts with the lowest energy state (ground state) and moves progressively from there up the energy scale until each of the atom’s electrons has been assigned a unique set of quantum numbers. It is the Pauli exclusion principle that requires the electrons in an atom to occupy different energy levels instead of them all condensing in the ground state. In the periodic table, the elements are listed in order of increasing atomic number Z. The number of electrons in each element’s electron shells, particularly the outermost valence shell, is the primary factor in determining its chemical bonding behavior. The configuration of these electrons follows from the principles of quantum mechanics. The chemical properties of the atom are determined by the number of protons, in fact, by number and arrangement of electrons. See also: Atomic Number – Does it conserve in a nuclear reaction? Atomic Number and Chemical PropertiesĮvery solid, liquid, gas, and plasma is composed of neutral or ionized atoms. It is the electrons that are responsible for the chemical bavavior of atoms, and which identify the various chemical elements. In a neutral atom there are as many electrons as protons moving about nucleus. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. The nucleus is composed of protons and neutrons.
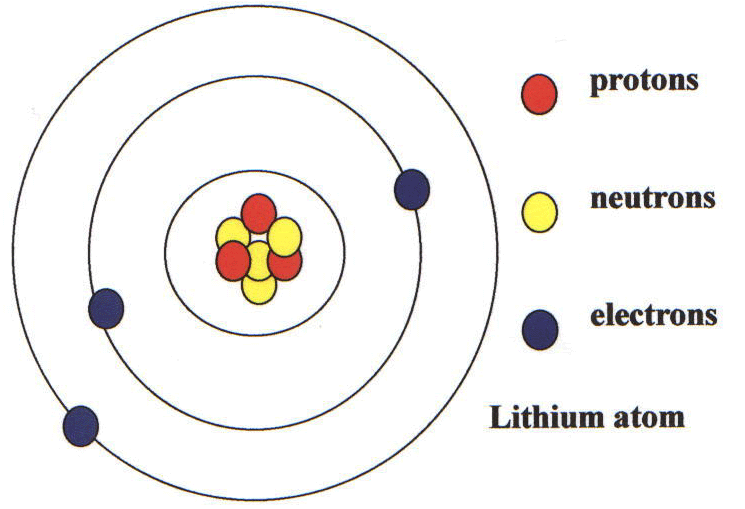
The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons.
